Reimagine TYK2 Inhibition
for Plaque Psoriasis
Unmet Needs in Plaque Psoriasis
Plaque psoriasis is a multidimensional, chronic inflammatory disease that extends beyond the skin, often requiring systemic therapy to achieve meaningful, durable disease control.1-3
Systemic-appropriate patients with plaque psoriasis need more effective oral therapies that address immune dysregulation.5-10
TYK2’s Role in Plaque Psoriasis
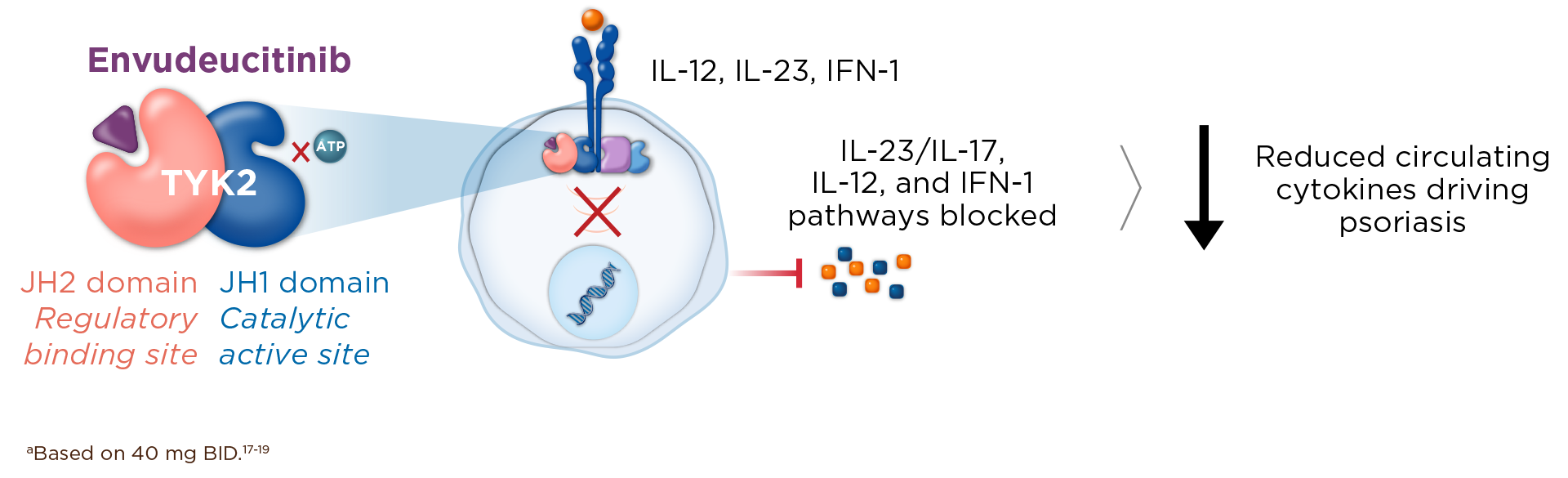
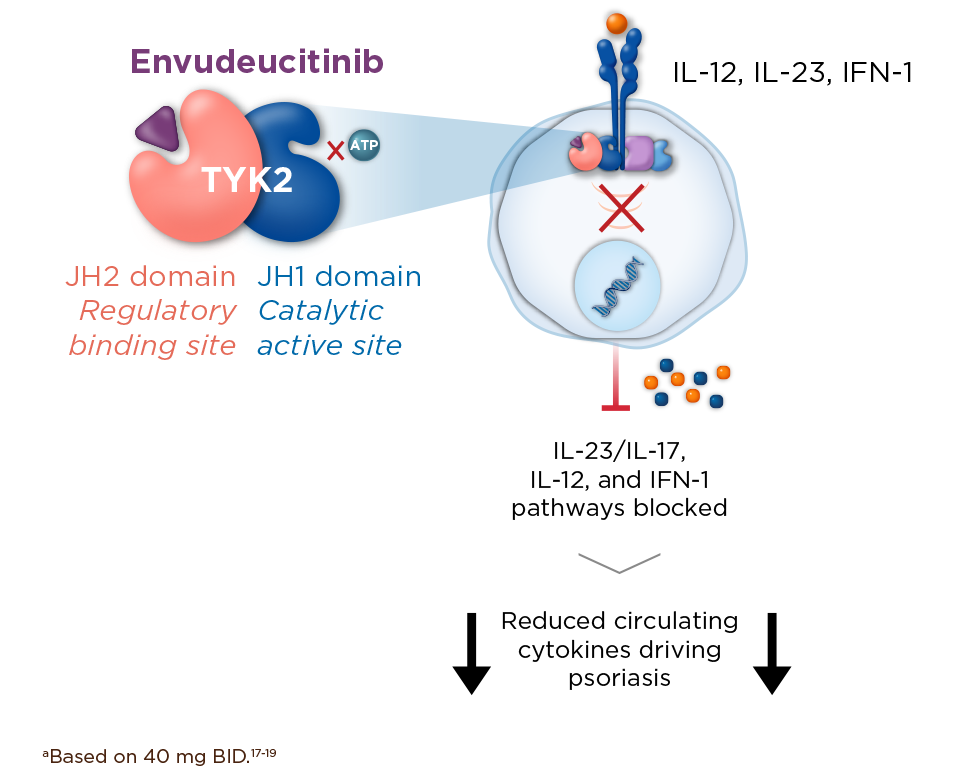
TYK2 is a central upstream mediator of multiple proinflammatory signaling pathways, including IL-23 and IL-17, that drive psoriasis pathogenesis and progression by fueling chronic inflammation, skin thickening, scaling, and itch, and systemic immune activation.11-16
Inhibiting TYK2 blocks both IL-23 and IL-17 to correct immune dysregulation.17-19
Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency.
Envudeucitinib is an Investigational, Next-Generation TYK2 Inhibitor
What is envudeucitinib?
What is envudeucitinib?
Envudeucitinib is an investigational, next-generation, highly selective oral allosteric TYK2 inhibitor, precision-engineered for maximal, 24-hour TYK2 inhibition.17-20,a


Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency.
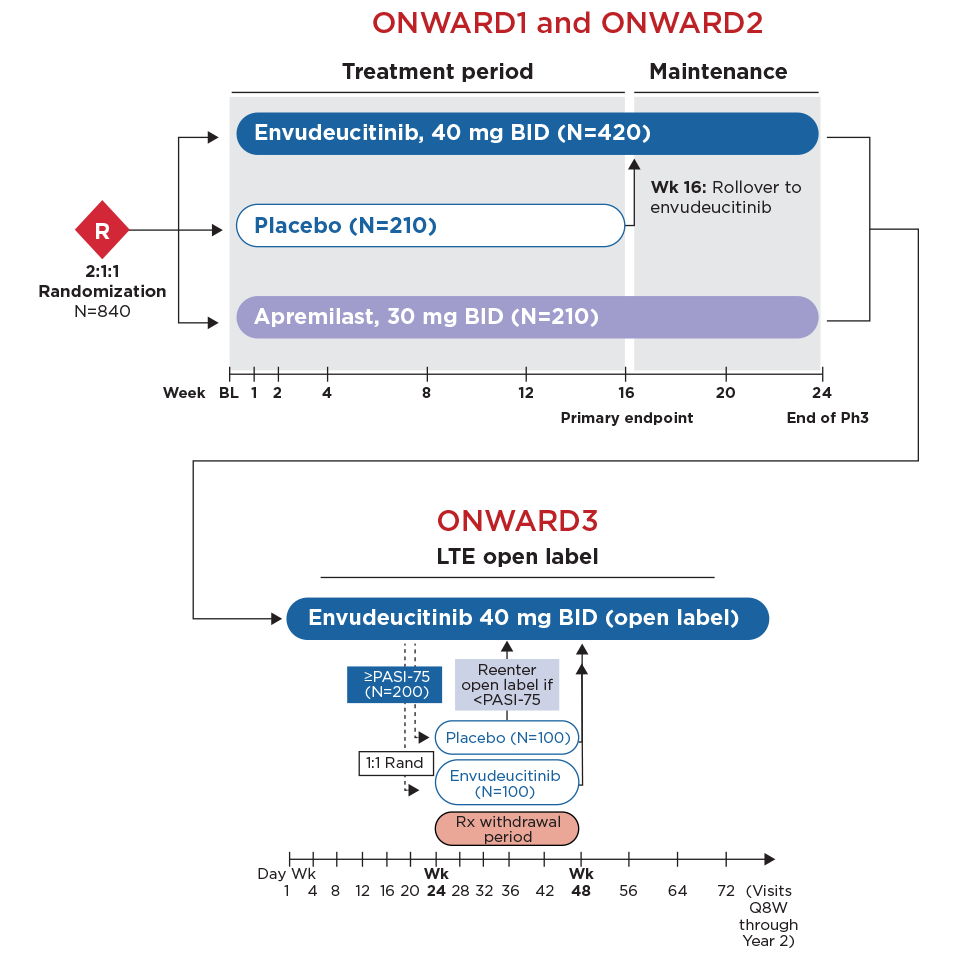
Latest Phase 3 results: ONWARD1 and ONWARD2
Envudeucitinib Phase 3 Data20
Reinforcing the potential of TYK2 inhibition for moderate-to-severe plaque psoriasis
In the global Phase 3 ONWARD1 and ONWARD2 trials, envudeucitinib met all primary and secondary endpoints with high statistical significance
Envudeucitinib achieved superior skin clearance vs placebo (p < 0.0001) on PASI 75 and sPGA 0/1

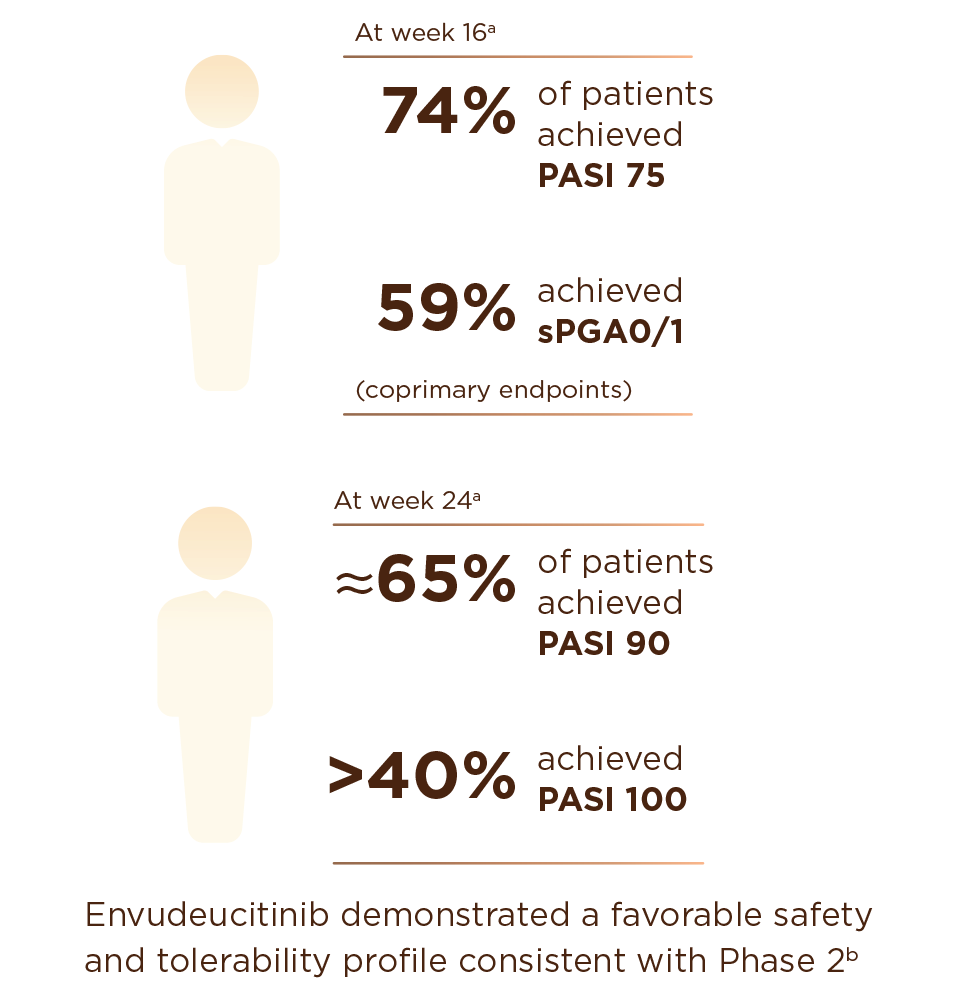
Early onset improvement was observed, with clear separation from placebo on PASI 90 as early as week 4
Consistent and clinically meaningful improvements across patient-reported outcomes relating to itch and quality of life were observed
Envudeucitinib also achieved superior skin clearance vs apremilast (p<0.0001) across all PASI endpoints at week 24
The most common TEAEs were headaches, nasopharyngitis, upper respiratory tract infections, and acne. No new safety signals were observed.
aOn average across both ONWARD1 (NCT06586112) and ONWARD2 (NCT06588738). bTreatment with envudeucitinib was generally well tolerated through Week 24 in both trials, with a safety profile consistent with Alumis' Phase 2 program, including the long-term extension trial. TEAE frequency and severity were similar across trials, with the majority being mild to moderate, transient, and responding to standard therapy, if required.
Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency.
AAD Late-Breaker: Phase 3 Results from ONWARD1 and ONWARD2
Reinforcing the potential of TYK2 inhibition for moderate-to-severe plaque psoriasis
Envudeucitinib (ESK-001) in moderate-to-severe plaque psoriasis: 24-week results from the randomized, double-blind, active comparator- and placebo-controlled, Phase 3 ONWARD 1 and 2 studies
Blauvelt A, et al. AAD 2026
Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency.
Phase 1/2 clinical trial results
Phase 1 data with clinical evidence of maximal TYK2 inhibition
Safety, tolerability, PK, and PD of the oral allosteric TYK2 inhibitor ESK-001 using a randomized, double-blind, placebo-controlled study design
Ucpinar S, et al. Clin Transl Sci 2024
PK, Safety, and Tolerability of ESK-001, an Allosteric TYK2 Inhibitor for Plaque Psoriasis: Evaluation in Asian Populations Compared to Caucasians
Noh Y, et al. AAD 2025
PK and PD Characteristics of ESK-001, an Oral Allosteric TYK2 Inhibitor, in Phase 1 Healthy Volunteer Trials
Ucpinar S, et al. AAD 2024
Phase 2 STRIDE and OLE studies data set through 52 weeks in moderate-to-severe plaque psoriasis
Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: Results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study
Blauvelt A, et al. J Am Acad Dermatol 2026
Safety and efficacy of envudeucitinib, a highly selective, oral allosteric TYK2 inhibitor, in patients with moderate-to-severe plaque psoriasis: results from the 52-week open-label extension period of the phase 2 STRIDE study
Papp KA, et al. J Am Acad Dermatol 2026
ESK-001, an Allosteric TYK2 Inhibitor, Modulates Disease and TYK2-Related Pathway Transcriptomic and Proteomic Biomarkers in Psoriasis STRIDE Trial Patients
Narayan NR, et al. AAD 2025
Patient-Reported Outcomes in the Phase 2 Studies of ESK-001, an Oral Allosteric TYK2 Inhibitor, in Adults with Moderate-to-Severe Plaque Psoriasis
Blauvelt A, et al. AAD 2025
ESK-001, a Highly Selective Oral TYK2 Inhibitor: 52-Week Phase 2 Study Results in Moderate-to-Severe Plaque Psoriasis
Blauvelt A, et al. AAD 2025
Achievement of Treat-to-Target Thresholds with Envudeucitinib, a Selective TYK2 Inhibitor, in Moderate-to-Severe Plaque Psoriasis: Results from STRIDE and OLE
Armstrong A, et al. Fall Clinical Derm 2025
Efficacy and Safety of ESK-001, a Highly Selective Oral TYK2 Inhibitor, in a Phase 2 Study (STRIDE) and OLE Study in Adults with Moderate-to- Severe Plaque PsO
Papp KA, et al. AAD 2024
Efficacy and Safety of ESK-001, a Highly Selective Oral TYK2 Inhibitor, in Moderate-to-Severe Plaque Psoriasis: Phase 2 Results Through Week 28
Blauvelt A, et al. EADV 2024
ESK-001, An allosteric TYK2 Inhibitor, Downregulates Biomarkers of Disease and TYK2 Activity
Tilley MK, et al. EADV 2024
Patient-Reported Outcomes in the Randomized, Double-Blind Phase 2 Study of ESK-001, an Oral Allosteric TYK2 Inhibitor, in Adults with Moderate-to-Severe Plaque Psoriasis
Blauvelt A, et al. EADV 2024
Exploratory Exposure-response (E-R) Analysis of ESK-001, an Allosteric Oral TYK2 Inhibitor, in Patients with Plaque Psoriasis
Ucpinar S, et al. EADV 2024
Transcriptomic Modulation in Psoriatic Skin Following Envudeucitinib Treatment: A Subgroup Analysis of a Phase 2 STRIDE Trial Skin Tape Strips
Zhou J, et al. Inflammatory Skin Disease Summit 2025
Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency.
What Is on the Horizon:
Clinical Development Milestones
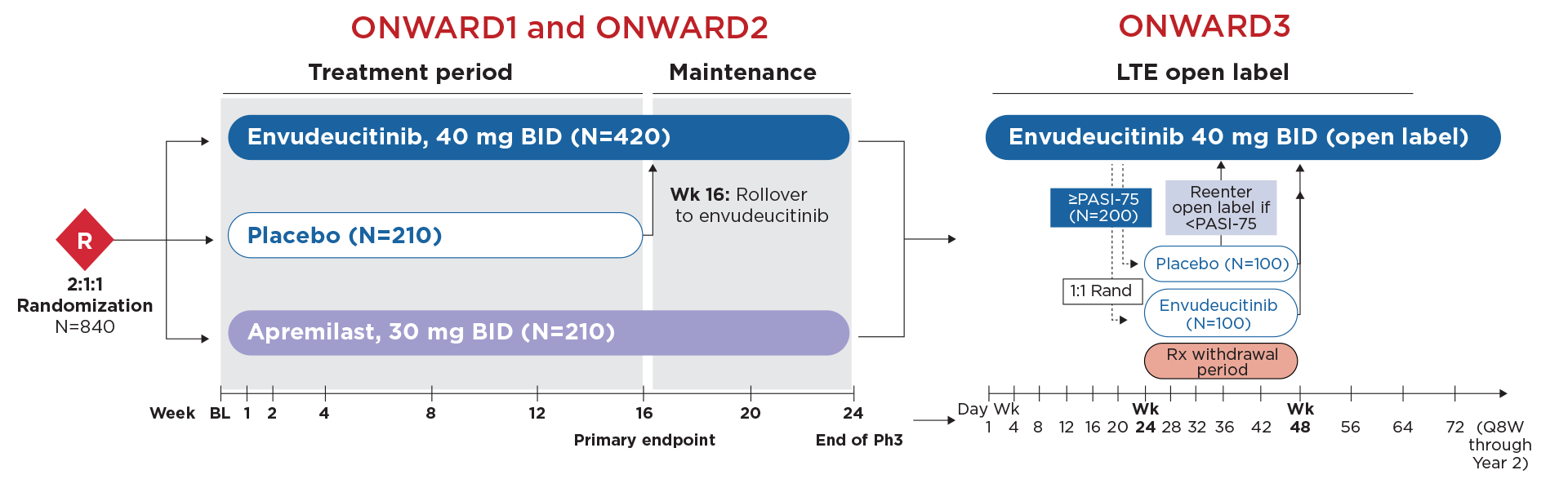
ONWARD3 long-term extension study is ongoing20
Patients who completed Week 24 of ONWARD1 or ONWARD2 were eligible to enter ONWARD3 (NCT06846541), an ongoing LTE study assessing durability, greater maintenance of response, and long-term safety of envudeucitinib in patients with moderate-to-severe plaque psoriasis. The ONWARD clinical trials did not have a fasting requirement.
Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency.
About Alumis
Alumis is pioneering a precision approach powered by data analytics and scientific insights to address immune dysfunction. Leveraging our deep understanding of TYK2-mediated pathways, we are committed to researching next-generation targeted treatments that can meaningfully transform patient outcomes.
Would you like to receive additional information and updates related to envudeucitinib and psoriasis?
Please provide your name, degree, country, and email.
[form placerholder]
By doing so, you consent to receiving communications via email.
AAD, American Academy of Dermatology; BL, baseline; BID, twice daily; JH, Janus kinase homology domain; IFN, interferon; IL, interleukin; LTE, long-term extension; MOA, mechanism of action; OLE, open-label extension; PASI 75/90/100, _75%, _90%, or 100% improvement from baseline in Psoriasis Area and Severity Index; Ph, phase; Q8W, every 8 weeks; QoL, quality of life; Rx, treatment; sPGA 0/1, static Physician’s Global Assessment of clear (0) or almost clear (1); TEAE, treatment-emergent adverse event; TYK2, tyrosine kinase 2.
1. Armstrong AW, et al. Nat Rev Dis Primers. 2025;11(1):45. 2. Menter A, et al. J Am Acad Dermatol. 2019;80(4):1029-1072. 3. Solmaz D, et al. Clin Exp Rheumatol. 2020;38(2):257-261. 4. Korman NJ. Br J Dermatol. 2020;182(4):840-848. 5. Lebwohl MG, et al. Dermatol Ther (Heidelb). 2022;12:61-78. 6. Schmidt L, et al. Dermatol Ther (Heidelb). 2023;13:2019-2030. 7. Smith SD, et al. J Dermatolog Treat. 2024;35:2339440. 8. Stein Gold L, et al. Presented at: Fall Clinical Dermatology Conference; October 23–26, 2025; Las Vegas, NV. 9. Strober B, et al. J Psoriasis Psoriatic Arthritis. 2025;10(1):22-27. 10. Strober B, et al. J Am Acad Dermatol. 2020;82(1):117-122. 11. Jensen LT, et al. EBioMedicine. 2023;97:104840. 12. Martin G. Dermatol Ther (Heidelb). 2023;13:417-435. 13. Gri_ths CEM, et al. Lancet. 2021;397(10281):1301-1315. 14. Singh R, et al. Int J Mol Sci. 2021;22(23):12793. 15. Armstrong AW, et al. JAMA. 2020;323(19):1945-1960. 16. Krueger JG, et al. J Am Acad Dermatol. 2022;86(1):148-157. 17. Blauvelt A, et al. J Am Acad Dermatol. 2026;94:57-65. 18. Papp KA, et al. J Am Acad Dermatol. 2026;94:187-195. 19. Ucpinar S, et al. Clin Transl Sci. 2024;17:e70094. 20. Alumis’ envudeucitinib delivers leading skin clearance among next-generation oral plaque psoriasis therapies in phase 3 program. Alumis Inc.; Press release. January 6, 2026. Accessed March 10, 2026. https://investors.alumis.com/news-releases/news-release-details/alumis-envudeucitinib-delivers-leading-skin-clearance-among-next. 21. Transforming Immune-Mediated Disease Treatment with Precision Engineered TYK2 inhibitors Corporate Deck January 2026. Alumis Inc.; Accessed March 10, 2026. https://investors.alumis.com/static-files/63f3ad30-30a6-4a31-8382-91187e67af0f.
Envudeucitinib is an investigational therapy not reviewed or approved by any regulatory agency.
